Dr. Blanca Molins from The IDIBAPS, Hospital Clínic de Barcelona explains a 2020 research paper she co-authored that was published by Aging (Aging-US), entitled, “Activation of C-reactive protein proinflammatory phenotype in the blood retinal barrier in vitro: implications for age-related macular degeneration.”
Behind the Study is a series of transcribed videos from researchers elaborating on their recent studies published by Aging (Aging-US). Visit the Aging (Aging-US) YouTube channel for more insights from outstanding authors.
—
Good afternoon, my name is Blanca Molins and I will explain you a little bit about our work that we published in Aging, which was entitled, “Activation of C-reactive protein proinflammatory phenotype in the blood retinal barrier in vitro and the implications for age-related macular degeneration.” I work in a primary hospital in Barcelona, in Spain, which basically is a research academic hospital in which we do a lot of research, but it’s basically focused on the patient and what the pharmacoloy questions they have; they come to the lab and we try to solve these questions that they need to address in order to better treat their patients. In this regard, the ophthalmology department has been, for a long time, working with many retinal disorders.
We started to look at what was happening with this protein, the C-reactive protein, which is an acute phase reactant. Several years ago, it was already identified as a risk marker for AMD, Age-related Macular Degeneration. Patients with increased levels of this protein were at higher risk, in theory, to develop AMD. Based on our previous work in another completely different area, when I was working in the cardiovascular field that I was working already with C-reactive protein, we saw that it wasn’t totally a marker, but also upper inflammatory test proinflammatory phenotype, depending on the confirmation of the protein. We brought all this knowledge that we had from the cardiovascular system and tried to study what was happening with this proteins at the retinal pigment epithelium. Few years ago, we already saw that one of the confirmations of the CRP, the monomeric form was proinflammatory and it was using barrier disruption.
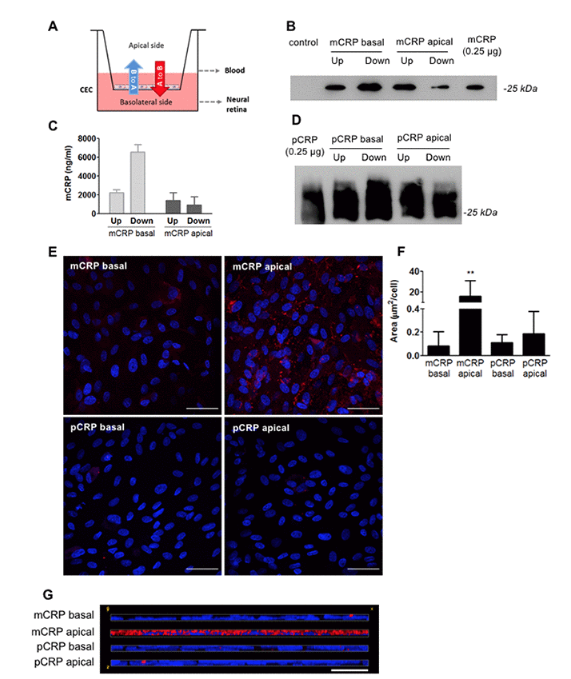
We did all this in a very simple in vitro system because we didn’t have much money to go for it in vivo. We had to find out more sophisticated way to study the role of this protein, but without doing in vivo studies, because we had very limited funding. We developed a blood retinal barrier model that was mimicking a bit more the real OBRb with co-culturing retinal pigment epithelial cells, to try to understand how this CRP was affecting the function of the blood retinal barrier. It was difficult because we wanted to really see how this CRP could reach the subretinal space, because there is no transcription of CRP in the RP. All our previous findings that we had seen that monomeric CRP could be proinflammatory in the retinal epithelium. If there was no physiological relevance in the sense that this protein could not reach the BRP, all our previous work had no sense.
In this work, we basically try to understand how this protein could reach the separational space. We saw that it was able to cross endothelial cells and deposit lower in the BA of deposits of the retinal P epithelium and we also were able to describe that this form of CRP, which is the one that pro-inflammatory could generate by in the surface of the RP, when this RP was damaged because of the surface present in the surface of damaged RP. Then, basically, we were able to show some relevance of our previous results, because we really were able to show that this CRP and especially the monomoy form, which is the draw inflammatory one can reach the sub RP space and can cross the grown endothelium and it’s able to disassociate locally. With these results, we are able to really go farther and really try to see how this CRP really has a relevant role in AMD in the progression of the disease.
Now what we will try to do next … in fact, we are working with that is trying to test in an animal model, whether the intravitreal or intravenous injection of the different forms of CRP can induce some damage in the retinal barrier and the ultra blood retinal barrier. With that, we’ll probably close up the loop and really tell us whether it makes sense to target on and go farther and study how this protein works in the pathophysiology of AMD. Then depending on the results we have, then maybe we can really consider that as a new potential target for AMD, or just seeing it as a bystander of the disease.
Click here to read the full study published by Aging (Aging-US).
WATCH: AGING VIDEOS ON LABTUBE
—
Aging (Aging-US) is an open-access journal that publishes research papers monthly in all fields of aging research and other topics. These papers are available to read at no cost to readers on Aging-us.com. Open-access journals offer information that has the potential to benefit our societies from the inside out and may be shared with friends, neighbors, colleagues, and other researchers, far and wide.
For media inquiries, please contact [email protected].